In this article, we will learn the basic concept of the iron-carbon phase diagram or equilibrium diagram. It is also called an iron carbide phase diagram as well. Let’s explore!
Iron-Carbon Phase or Equilibrium Diagram
Iron-Carbon Phase Diagram Basics
Carbon is the most crucial member of alloying elements which notably affects the allotropy and chemical as well as mechanical properties of iron. Iron is known as an allotropic material, the allotropic changes that take place in iron are governed by alloying elements, and the crucial material is iron which causes most of these changes.
- Alloy metals do exist in various phases.
- Phases are known to be physical homogenous states of an alloy; these alloys have a very explicit chemical composition.
- This structure of atoms has various properties in different phases.
- We can even choose the phase we need; we can use it in our required application.
- Though only some special alloys can actually exist in various multiple phases.
- We can use these metals and their alloys in different phases by heating them to specific temperatures using heat treatment procedures.

That’s the reason why the study of this system is very crucial to understand the behavior of steel and cast irons and explore its more basic features as well as the most complex ones. This system is influencing the complex microstructure of alloys one way or another and also changing their mechanical properties.
Steel has some accidental alloying elements which change the stigma of this diagram, but its changes should be performed cautiously because for many engineers this diagram acts as a guideway.
What is Phase or Equilibrium Diagram?
Phase Diagram Basics
Let’s say, for example, the phases which we found in the simple carbide system preserve in complex steels, but it is essential to examine the outcomes these alloying elements have on the inception. The iron-carbon or iron-carbide phase diagram provides a valuable substratum and allows us to construct knowledge of both carbon and steel in their massive variety.
Phase or Equilibrium Diagram Definition
Equilibrium Diagrams are graphical representations of the phases which we can call different chemical compositions present in an alloy at different temperatures.
- The diagram explains the appropriate conditions for two or more two phases that exist in the same equilibrium.
Let’s say, for example, the water phase diagram tells us more about the point (also known as a triple point) where water coexists in 3 different phases at the same time.
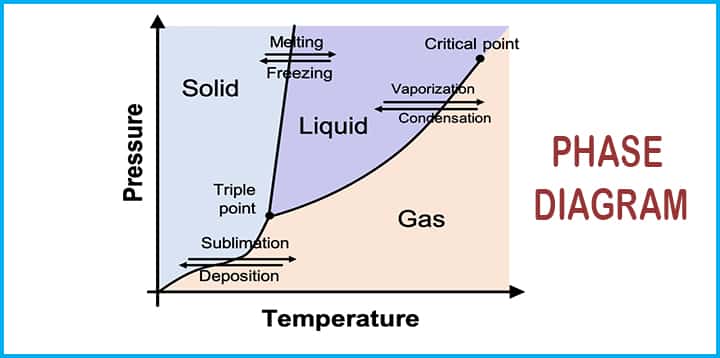
- It may be noted that this diagram is not truly an equilibrium diagram as the equilibrium word indicates no change of phase with time. As we know, the compound of iron carbide degraded into iron and carbon, but even this type of decomposition takes several years to complete.
- Iron carbide equilibrium diagram shows us metastable conditions, one can achieve equilibrium changes under slow heating and cooling.
Importance of Iron Carbon Equilibrium Diagram
- According to the requirements of different applications, we can develop new alloys by handing them at different temperatures and changing the compositions of carbon in it.
- For the improvement of the various properties of alloys such as chemical, physical and mechanical, we can evolve and control relevant heat treatment strategies.
- Ultimately upgrading product monotony by troubleshooting the difficulties arising during the application of these alloys.
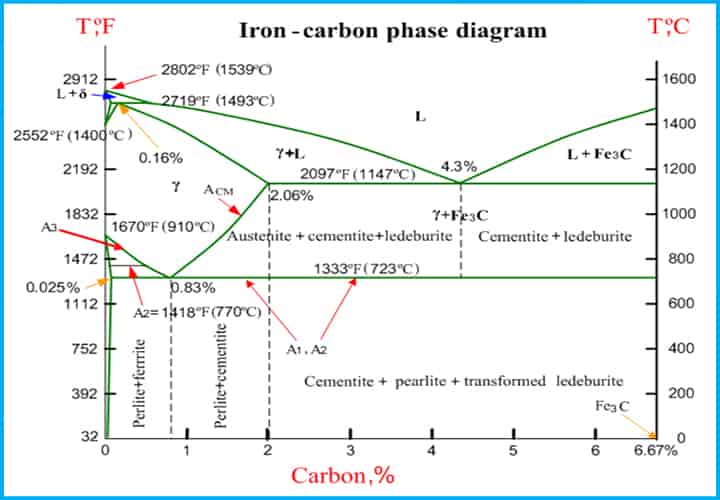
Critical Temperature Lines
This is the temperature at which phase changes occur during heating or cooling.
- In the above diagram, it can be seen various lines titled A1, A2, A3, and ACM. These boundaries are actually considered as phase changing points as the temperature of metal decreases and increases, phase changes occur.
- Usually, when heating a substance, an increase in temperature occurs. But when we go according to these lines, it resulted in a change in the chemical composition of the structure into a different phase, and, therefore, the temperature increment stops until the whole phase transformation occurs. This phenomenon of changing the phase without an increase in temperature is known as “Temperature Arrest”.
- By using different alloy steels, Boundaries on the diagram change in direction, either it decreases or it rises.
- The rate of heating and cooling of compositions have a certain effect on the boundary lines. When the rate of heating or cooling is slower than two consecutive lines, they are nearer to each other, so even though the inconsistent rates of cooling and heating will occur at some temperature.
In the phase system, certain symbols are used to denote the critical temperature.
- Ac = arrest during cooling
- Ar = arrest during heating
- A0 = This is the critical temperature of cementite i.e. 210° C.
- A1 = It is the lower critical temperature point where austenite to pearlite transformation occurs .
- A2 = It is the critical temperature (768° C )of iron where it turns into a paramagnetic substance. It is also known as Curie temperature.
- A3 = It is the boundary temperature between γ ferrite and (austenite + ferrite).
- Acm = It is the boundary temperature between γ ferrite and (austenite + cementite).
- A4 = At this temperature α-ferrite turns into δ-ferrite.
Eutectic Point
- As we can see on the equilibrium diagram, boundary lines meet with each other at some point and this point is known as the Eutectic point. The inception of these points is totally accidental. In the mixture of two solid phases, the liquid phase freezes and that’s how the eutectic reaction takes place.
- In simpler words, it is known as the lowest temperature at which liquid exits in the system. The incepted alloys formed at these eutectic points are known as eutectic alloys.
- The alloys are called eutectic alloys formed at this point.
Micro-Constituents in Iron-Carbon Phase Diagram
Different phases that occur on the iron-carbon phase or equilibrium diagram are discussed below.
Cementite or Iron carbide
To form cementite or iron-carbide, we need to have a fixed amount of carbon and a fixed amount of carbon, and that’s the reason why this is known as the metastable phase of this alloy.
- It contains 6.67% of carbon, and it is an extremely hard and brittle compound having very low tensile strength.
- This alloy decomposes into iron and carbon at room temperature, but the decomposition rate is very slow. The decomposition rate for this alloy can increase by adding some elements and can be provided, for instance, temperature rise.
- This alloy is the hardest structure that appears on the iron-iron carbide diagram. This property is suitable for various applications such as the strengthening of steel. The mechanical properties of such alloys depend upon how we mix them with the ferrite.
- It has a complex orthorhombic crystal structure ( i.e. 12 iron atoms present with 4 carbon atoms in a unit cell.).
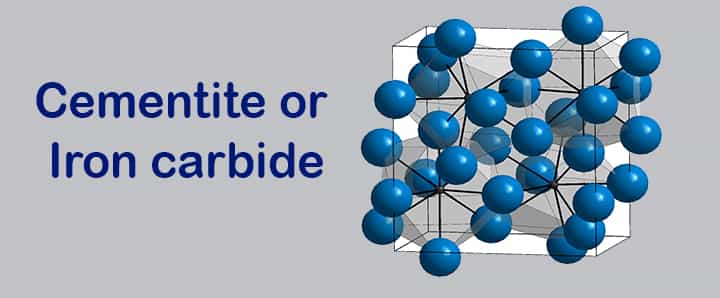
Why is cementite considered a metastable phase?
Cementite/Fe3C is a compound of iron and carbon, which has a weight of 6.67% by carbon. If we will heat this compound and keep it at a constant high temperature (650-700 degrees Celsius) for a long time, it will decompose into graphite and ferrite. For this momentary stability nature of cementite, it is considered a metastable phase.
Austenite
Austenite is a solid solution of carbon in Face Centered Cubic with having a solubility of 2.14% carbon at 2065°F.
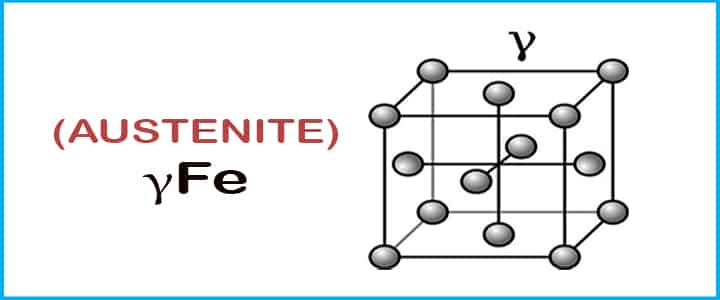
This is also known as γFe.
- When we heat this solution, it converts into ferrite at a temperature above 2543°F.
- Austenite is usually unstable at temperatures below 727°C.
- This phase is non-magnetic in nature, and it changes rapidly.
- Austenite is very soft, and it possesses mechanical properties such as ductility and malleability, and it is also paramagnetic.
- Above 1100°C sheets of steels are usually rolled when they are in an austenitic state because of their ductility and malleability properties.
- The Tensile strength of this solution is around 150000 psi.
Ledeburite
Ledeburite is a mixture of austenitic alloy and cementite.
- It does have 4.3% of carbon, and it is created at 1129.
- When carbon is more than 2%, it exists in phase, and it is represented by the dividing line between two materials, and those materials are cast iron and steel.
Ferrite
Ferrite is a solid solution where a small amount of carbon is dissolved in iron which has a body-centered cubic crystal structure. This phase of solution only exists at very high temperatures only.
is a solid solution where a small amount of carbon is dissolved in iron which has a body-centered cubic crystal structure. This phase of solution only exists at very high temperatures only.
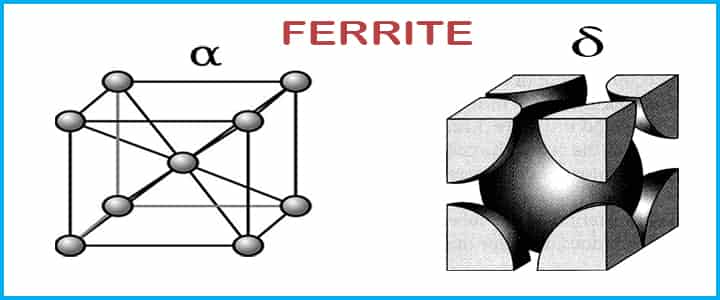
- This solution has a melting point of 1538°C. Ferrite is the softest structure on the iron-iron carbide diagram.
- Ferrite acts magnetically at low temperatures, but its magnetic properties are dethatched once its temperature rises and above 786°C temperature it becomes non-magnetic.
- This solid solution draws a microstructure of ferrite showing fine grains, each grain has a different arrangement in each atom.
- α-ferrite : It is an interstitial solid solution of carbon in BCC iron which is stable in between -273° to 912° C. The maximum solubility of carbon is 0.02 weight % at 727° C.
- δ-ferrite : It is an interstitial solid solution of carbon in BCC iron.
- It is stable between 1394°-1538° C. The maximum solubility of carbon is 0.09 weight % at 1538° C.
Pearlite
Pearlite is a solution that contains about 0.80% carbon, and it is formed at 1333°F, and it requires very slow cooling. It is a sort of lamellar mixture of solution ferrite and cementite. Thin plates of cementite with a white matrix complete the structure of the pearlite.
- It has a tensile strength of about 120000 psi. This particular solution precisely needs a fixed amount of cementite and ferrite.
- If the amount of carbon required is less than that of 0.80%, the iron and carbon come together to form the Fe3c compound until all the carbon is dissolved completely. With the required amount of ferrite with the cementite, one can form a pearlite solution.
- The ferrite which remains in pearlite is known as free ferrite, and that’s how pro eutectoid ferrite is formed. The steel solution which has even some amount of pro eutectoid ferrite is known as hypo eutectoid steel.
- In the grain boundaries, there is excess cementite present. This excess cementite can be called pro eutectoid cementite.
Graphite
It is hexagonal in the structure where sheets of primary bonded carbon and secondary bonded carbon atoms hold together.
Check a very nice video from IIT Kanpur,
Mechanical Properties of Steel affected by Carbon
- In general, we know that the mechanical properties of any solution rely on the different phases of materials, and these phases of materials are re-arranged in such a way that they make up the structure.
- As we studied earlier about the two materials, those are ferrite and cementite. These two materials have relatively low tensile strength, but the difference between them is that ferrite is soft in nature, but on the other hand, cementite is very hard. But we can change these properties of ferrite and cementite by forming them together as pearlite. It will also give us the advantage of high tensile strength which they both lacked individually.
- As we discussed earlier, mechanical properties can be changed by changing their chemical compositions. We use this property on hot-worked steel. For the hypo eutectoid steels, the amount of perlite increases with an increase in the carbon content.
- By increasing carbon content, the strength and hardness will also go up. The only drawbacks we faced during this transformation are ductility and impact strength of the material decreases since we increased the carbon composition.
- At a certain point above the eutectoid composition, the strength of the material falls off and shows some brittle properties, but Brinell hardness resumes to increase and it hardens the cementite.
Invariant reactions
There are three types of invariant reactions which occur in the iron-carbon system.
Peritectic reaction
It occurs at 1495° C. 0.09% of δ-ferrite reacts with 0.53% of liquid and produces 0.17% of Austenite/ γ-ferrite. Here, BCC iron (δ-ferrite) turns into FCC iron (Austenite). During cooling, the molten steel starts solidifying and the first solid, which is going to nucleate, is δ-ferrite. More amount of δ-ferrite solidifies with the continuation of cooling. We can use the “Lever rule” to calculate the amount of δ-ferrite in fraction.
Reaction: δFe + Liq –> γFe
Eutectic reaction
It occurs at 1146° C. Fe- 4.3% C alloy is called eutectic cast iron. During the cooling of cast iron, the lowest melting liquid decomposes into two solid phases. This mixture of two solid phases (Austenite and Cementite) is also known as “Ledeburite”.
Reaction : Liq –> γFe + Fe3C
Eutectoid reaction
It occurs at 727° C. During the cooling, Austenite becomes a lamellar mixture of ferrite and cementite. This mixture is called the “Pearlite” mixture because of its pearly appearance under the optical microscope. Here, 0.77% of Austenite decomposes into 0.02% of α-ferrite and 6.67% of cementite.
Reaction : γFe –> αFe + Fe3C
Transformation of Slowly Cooled steels in Iron Carbon Phase Diagram
Hypo Eutectoid Steels
To understand the term Hypo eutectoid steel, we should consider relatively slow cooling of iron-0.4% Carbon steel under thermal equilibrium conditions from the temperature of around 1600°C to room temperature.
- On further cooling, more ferrite solution tries to form till the temperature of 1495°C.
- On more cooling, the liquid solution changes into solid austenite until a particular temperature is reached, and thus solidification completes.
Eutectoid Steels
When this solution cooled from 1600°C, it started solidifying at room temperature, which directly created into solid austenite, and at some point when this structure is in a microstate; this has only grains of austenite.
- Further, when we cooled it down to eutectoid temperature, no changes occurred in the solution.
- At 727°C, it gives pure pearlite which has undergone a eutectoid reaction.
Advantages of Iron Carbon Phase diagram:
- To understand the phase changes in iron and carbon alongside its alloying elements such as steel to attain a thermal equilibrium state.
- To gain the exact required mechanical properties with slight phase changes is possible.
- Slow cooling enables us to study the behaviour more accurately; also, it gives us an advantage of slow phase transformation.
- For engineering purposes, the diagram works as a pathway we need to follow for attaining required phases and required mechanical properties very rapidly and accurately.
Limitations of Iron Carbon Phase Diagram
- Equilibrium cooling rates are much slower than that of the actual heat treatment cycles; it is because of the different behaviour of the alloys at practical use.
- Due to these drawbacks, this has put a major setback on the interpretation of the diagram at lower temperatures.
Conclusion
The Iron-carbon phase or equilibrium diagram is an overall study of iron, carbon, and their alloys and how they act under the various equilibrium conditions. In this blog, we studied these aspects of the equilibrium diagram and also how phase transformation depends upon the temperature conditions.
Although it is known as an equilibrium diagram, the temperature is not stable at some phases, so it is also called a metastable diagram. Studying the microstructure of various solutions provides us with the details of their chemical compositions and their mechanical properties. However, in actual practice materials do not exactly act according to the diagram stated, but the difference between theoretical and practice is very little, so we can consider this diagram as a guide.
